How does it work?
Use of RESCAP for age-related diseases
RESCAP will be developed to treat different age-related diseases, such as Diabetes, Parkinson, Frailty and Alzheimer’s
Benefits
Why RESCAP?
1
Interferes with root-cause
RESCAP restores decreasing levels of iAP-enzyme activity. This decreased iAP level is a critical factor for the development of age-related diseases.
2
Fully natural
RESCAP is the naturally existing enzyme ‘intestinal alkaline phosphatase’ and therefore unlikely to cause drug-related side effects.
3
Safe
RESCAP can be dosed orally and does not enter the bloodstream, ensuring an optimal safety profile.
Invest in RESCAP
We are looking for a partner to invest in the development of RESCAP
How does it work?
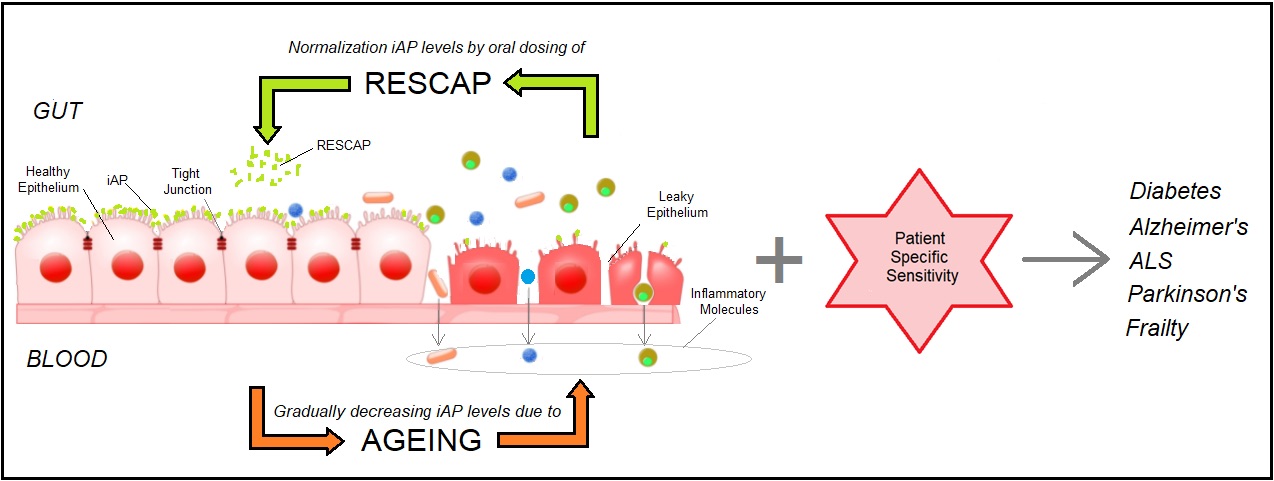
RESCAP treats age-related diseases by restoring the gut barrier
During ageing the gut-barrier gradually becomes more permeable as intestinal alkaline phosphatase (iAP) levels decrease. This leaky barrier causes inflammatory molecules to enter the blood which causes inflammation/immune activation. This inflammation/immune activation is a prerequisite for ageing and for the development of various age-dependent diseases. The exact age-dependent disease which develops in patients with a leaky barrier depends on the patients specific sensitivity, e.g. caused by mutations in their DNA, or by their life style. Interestingly however, these diseases only develop after the gut barrier gets leaky. By normalizing the normal iAP levels in the gut by orally dosing RESCAP the gut barrier closes again. Thereby RESCAP re-installs the pre-disease state, without the requirement to affect the patient sensitivities to the specific diseases they developed!
FAQ
Frequently asked questions about RESCAP
RESCAP is our proprietary name for the enzyme alkaline phosphatase (AP) dosed as a drug. Orally dosed RESCAP consists of the intestinal type of Alkaline phosphatase which is responsible for keeping the gut-body barrier closed, which prevents low grade inflammation and ageing in various animal models.
As RESCAP is a natural alkaline phosphatase enzyme that is normally present in a health gut, the gut will not be exposed to unnatural conditions by dosing RESCAP. Furthermore, RESCAP is not taken up by the body, but active inside the gut. As no internal exposure of organs occurs after dosing RESCAP, damage to these organs is unlikely.
Indications with a small total number of patients can be treated with RESCAP after scaling up of its production. However, once RESCAP would be used for bigger age dependent diseases and possibly even ageing, large scale production will be required. AMRIF has plans ready and pilot studies done to accomplish such a large scale production
“Drugs” are man-made molecules often selected from a large number of man-made compounds using in vitro and in vivo screenings assay’s. The disadvantage of man-made drugs is that they can have unpredictable side effects or metabolites, which can cause toxicity. “Biologicals” are a particular type of drug, obtained from animals and selected by evolution for an optimal effect. When used at levels which are normally present in vivo they generally show less unpredicted toxicity.
Drugs can either combat symptoms of diseases or treat the cause of the disease. The latter types of drugs are called “biological response modifyers”. RESCAP is a “biological response modifyer” as it takes away gut-induced inflammation, which is a pre-requisite condition for the development of age-related-diseases
No, RESCAP is not on the market yet. AMRIF is working towards a quick registrational approval program for RESCAP for clinical use and is searching funding for accomplishing that.
At the current small scale of production, RESCAP is an expensive product. However, pilot studies have shown that increasing the production volumes is feasible, which is expected to affect the production costs of RESCAP considerably.
